Effect of Obesity on semen parameters
Nagashree R¹, Praveena M²
¹Professor &Head of the Department, ²Final year PG, Department of Physiology,
PSG Institute of Medical Science & Research, Coimbatore, TamilNadu, India
Abstract
Background: Obesity is a non-communicable disease and has grown to pandemic in recent years in western as well as in developing countries. It should be considered as an etiology of male infertility, as there is an increase in incidence of overweight and obesity in men of reproductive age.
Aim: To study the effect of obesity on semen parameters.
Materials and methods: This was a cross sectional study, which involved 75 male participants, aged between 20 to 50 years. Paternal height and weight, life style habits and semen analysis data were collected from medical records of fertility department. Men were divided into three groups based on their body mass index (BMI). Group1 (n = 25)- Normal (18.5 -24.99 kg/m2), Group 2(n = 25) – Overweight (25.0 -29.99 kg/m2), Group 3 (n = 25)- Obese (BMI>30 kg/m2).Semen parameters were analysed by applying WHO 2010 criteria as normal cut off values. The data obtained was analysed using logistic regression.
Result: This study showed statistically significant association between obesity and abnormal sperm concentration, low sperm motility, low sperm vitality with an adjusted odds ratio of 26.36 (3.05 –227.74),14.34 (1.18–174.00),8.59 (2.34 – 31.49) respectively after adjusting for age, smoking habits, intake of alcohol and physical activity.
Conclusion: This study finding strongly indicates that obesity has an association with semen parameters. In future, it is needed to work in that direction to reduce such factors, which can affect the fertility of males.
Keywords: Body mass index (BMI), Male infertility, Obesity, Semen parameters.
Corresponding Author ::
Dr.Praveena. M,
Final year PG in M.D Physiology, PSGIMS&R.
Door No 204, Sneham Apartments,
Annanagar 1st Main Road,
Peelamedu, Coimbatore.
Telephone: +91 9566371859
E-mail: [email protected]
Introduction: Obesity is a global health problem not only in Western countries but also in developing countries like India.1 American Medical Association (2013), recently classified obesity as a disease. It affects not only cardiovascular system but also has a role in many other related health disorders. It accounts for 7.5% of the total burden of disease. In India, the prevalence of obesity is 12.6% in women as compared to 9.3% in men.2The rate of obesity in reproductive-age men has nearly tripled. Obesity reaches epidemic proportions with 1.6 billion adults being classified as overweight and an extra 400 million adults classified as obese.3Obesity adversely affects male reproduction by endocrine, thermal, genetic and sexual mechanisms.3It has also been reported that there are considerable numbers of overweight and obese infertile couples in their reproductive years and infertility affects approximately 15% of couples of which 40% is attributed to the male factor.4,5 The International Committee defines infertility for Monitoring Assisted Reproductive Technology and WHO as the ‘failure to achieve a pregnancy after 12 months or more of regular unprotected sexual intercourse’. WHO estimates the overall prevalence of primary infertility in India to be between 3.9% and 16.8% with regional variation.Among the Indian couples seeking treatment, the male factor is the cause in approximately 23%.6,7 Semen analysis is an important part of male fertility assessment (WHO, 2010).The relationship between obesity and male fertility has been largely ignored until recently. The relationship between exposure to lifestyle factors like consumption of alcohol, smoking, coffee, tea intake, occupation, physical activity and adverse effects on male reproductive health is debated in the scientific literature and these controversies have increased public and regulatory attention. With this background, this study was aimed to study the effect of obesity on semen parameters.
Study design: Cross sectional study.
Study Population: Males between 20 to 50 years of age attending the Fertility department.
Study Duration: Study Duration:August 2018 to July 2019.
Sample Size: 4pq/d2 = 334.Convenient sample e-75.
Inclusion criteria: Study participant males aged between 20 to 50 years.
Exclusion criteria: Males aged <20 and >50 years and those with history of varicocele, hydrocele, orchiditis, cryptorchidism, abnormal location of the testes, torsion of the testis, severe trauma to the genitalia, testicular tumors, genetic abnormality, azoospermia,vasectomy, orchidopexy and hernia surgery were excluded from the study.
Materials: Semen samples were collected by masturbation into a sterile plastic container in fertility department and men were instructed of three to five days of abstinence before the analysis. Semen parameters was analysed by conventional method in Department of Fertility.
Methodology: The study was conducted in the Department of Fertility, after getting clearance from the Institutional Human Ethics Committee (IHEC).Data like personal habits like consumption of alcohol, smoking, occupation, physical activity, height and weight were obtained from the medical records of males between 20 to 50 years of age from March 2017 to March 2019. Based on the inclusion and exclusion criteria study total 75 male participants were divided into three groups based on their body mass index(BMI). The body mass index (BMI) calculated as weight in kilograms divided by the squared height in meters.Group1 (n = 25)– Normal(18.5 -24.99 kg/m2), Group 2(n = 25) – Overweight (25.0 -29.99 kg/m2), Group 3 (n = 25) – Obese (BMI>30 kg/m2).8 World Health Organization 2010 criteria were used as cut off for semen analysis.9 The semen parameters like semen volume assessed by graduated tube, spermatozoa concentration, motility and progressive motility determined by Makler counting chamber. Sperm vitality assessed using eosin and nigrosine reagent and morphology data were determined microscopically. Semen analysis data were obtained from the records in Department of Fertility. Data thus collected were subjected to statistical analysis using an SPSS software version 24.0.
Table: 1 Association between semen parameters and body mass index Parameters Body mass
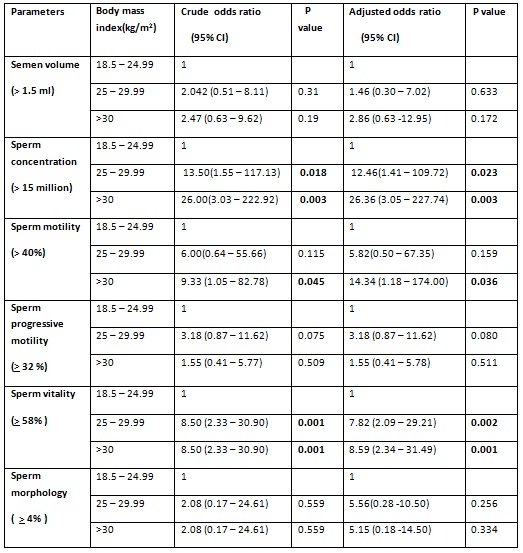
CI = confidence interval.
Logistic regression used to adjust for age, smoking, alcohol intake, physical activity.
p – value< 0.05 and < 0.001 = statistically significant.
Results: In this study, data was analysed using logistic regression.
Analysis of semen parameters (Table: 1) was done by applying WHO 2010 criteria as normal cut off values. This study showed statistically significant association between obesity and abnormal sperm concentration after adjusting for age, smoking habits, intake of alcohol, physical activity with an adjusted odds ratio of 26.36 ( 95% CI 3.05 – 227.74).
Obese men showed low sperm motility with an adjusted odds ratio of 14.34 (95% CI 1.18–174.00) and statistically significant after adjustment for confounders. This study also showed statistically significant association between obesity and semen parameters like volume were low and morphology were abnormal in obese compared to normal weight men but showed no statistical significance.
Discussion: The word obesity has been derived from ‘obesus’, which is a Latin word, meaning ‘one who has become plump by eating.’10The relationship between obesity and alteration of sperm parameters is multifactorial due to alterations of the hypothalamic pituitary gonadal (HPG) axis being involved in this process.
Lifestyle factors such as weight management, cigarette smoking, alcohol and exercise, may affect fertility. Smoking causes damage to the integrity of sperm DNA by increasing amounts of seminal leucocytes, which leads to increased reactive oxygen species generation. It has been linked with the inflammatory response induced by the metabolites of cigarette smoking. Smoking also affects endocrine function by increasing serum levels of both FSH and LH and decreasing testosterone.11,12Alcohol consumption has been thought to cause testicular atrophy, decreased libido, and decreased sperm count. An association of alcohol and sperm morphology has also been noted. Alcohol seems to have a large impact on both sperm morphology and sperm motility. Oxidative stress has been found to increase with alcohol consumption. A healthy amount of exercise in men can be beneficial. Physically active men exercising for at least three times a week for one hour had an increase in almost all sperm parameters in comparison to men who participated in more frequent and rigorous exercise. Diet combined with exercise in obese male had significantly better sperm morphology, total motile sperm counts, sperm concentration and increased sperm motility. It also decreased both sperm DNA damage and reactive oxygen species.
In this study, obese men and overweight were found to have abnormal sperm concentration when compared to normal body mass index men similar to the study conducted by Hammond AO et al.10 In obese men, aromatization of steroids to estrogens in peripheral tissues leads to the hypogonadotropic hyperestrogenic hypogonadism causing a significant decrease in total and free testosterone levels and increase in estradiol, both of which has deleterious effects on spermatogenesis. There is a decrease in sex hormone-binding globulin, most commonly mediated by hyperinsulinemia, emphasizing the negative feedback effect of elevated total estradiol levels. Obesity causes the endorphins to be increased, thus leading to both lower LH pulse amplitude and GnRH production. Obesity may directly alter spermatogenesis and Sertoli cell function, by decreasing inhibin B levels when compared with the decrease of FSH.13One of the common abnormal sperm parameter noted in obese males is DNA fragmentation. It indicates a poor quality of overall spermatogenesis, by affecting the concentration and motility of sperm.14The process of spermatogenesis is highly sensitive to heat, with optimal temperature ranging between 34–35°C in humans. Increased testicular heat is associated with reduced sperm motility, increased sperm DNA damage and increased sperm oxidative stress.
Moreover, this study also showed statistically significant association between obesity and low sperm vitality, but there was no statistically significant association to sperm volume, altered morphology in obese men compared to the study done by Pauli EM et al 16 which showed no relation between sperm vitality, volume, morphology, in obese men. Similarly, the study conducted by Beloc et al17 showed no statistically significant relationship between obesity and sperm morphology.
The study conducted by OboseRufuset al 18 showed a negative association between BMI and sperm morphology. Evaluation of the sperm morphology may be disputable since it continues to be affected by the influences of the subjectivity of the observer and because of the lack of objective measurement standards.
Serum inhibin B levels have been demonstrated to be a sensitive marker of spermatogenesis, indicating quantity and functionality of seminiferous tubules. Because there was no significant association of testicular volume with BMI, the lower concentrations of inhibin B in obese males seems to indicate decreased tubule function not yet resulting in decreased tubular volume. The effects of obesity on adult male reproductive health are primarily mediated through disruption of hormonal activity in the hypothalamic pituitary gonadal (HPG) axis, as well as physical effects of excess fat and increased scrotal temperature. Excessive visceral fat, more so than subcutaneous fat, leads to increased oestradiol levels due to increased activity of aromatase, which is produced by adipose tissue and converts testosterone to oestradiol, thus deregulating the HPG axis.
Other mechanisms that may contribute to obesity effects on male fertility include an increased release of adipokines from adipose tissue; and increased scrotal temperature, due to increased fat deposition in the upper thighs and abdomen, which interferes with spermatogenesis. Adipose tissue is an active endocrine organ. It produces adipokines, like leptin, and proinflammatory cytokines, like interleukin-6 (IL-6), which can affect spermatogenesis and sperm functioning. An increase in IL-6 levels leads to decreased ovum penetrating capability of spermatozoa and produces high levels of damaging reactive oxygen species, which disrupts the penile endothelium.
Leptin, a hormone responsible for satiety, also influences male fertility both at the level of the testes and at the HPG axis. An increase in fat mass leads to increased leptin levels and show an inverse relationship with serum testosterone levels in overweight and obese people. High leptin levels have a negative influence on testosterone synthesis from Leydig cell by inhibiting the conversion of 17-OH progesterone into testosterone.
Conclusion: This study supports that there is an association between obesity and altered semen parameters. Preventing obesity is key to abolish its negative impact on semen parameters. The need to look into the factors, which are causing a rise in male infertility, and attempts, should be made to control such factors in the near future.
Limitations and recommendations: The sample taken for this study was small to demonstrate the association between obesity and altered semen parameters. Therefore, more studies with larger sample size are needed to study the effect of obesity on semen parameters. This study throws light to create awareness about male infertility in society and to reduce the future male infertility. Further studies need to concentrate into underlying etiology, benefit of weight reduction among obese and treatment of male infertility.
Acknowledgements: Nil.
Conflict of Interest: Nil.
References:
1. Stefan N, Haring HU, Schulze MB. Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinology 2013; 1: 152-62.
2. Nagendra K, Nandhini C et al. A community based study on prevalence of obesity among urban population of Shivamogga, India. Int J Community Med Public Health. 2017 Jan;4(1):96-99.
3. Nguyen DM, El-Serag HB. The epidemiology of obesity. Gastroenterology Clinic North America. 2010; 39:1–7.
4. PasqualiR, Patton Gambineri A. Obesity and infertility. 2007; 14:482 -7.
5. Guney AI, Javadova D, Kirac D, Ulucan K, Koc G, et al. Detection of Y chromosome micro deletions and mitochondrial DNA mutations in male infertility patients. 2012; 11(2): 1039-48.
6. International Committee for Monitoring Assisted Reproductive Technology and the World Health Organization (WHO) revised glossary of ART terminology. Fertility Sterilization.2009; 92:1521524.
7. MacDonald A, Herbison G. The impact of body mass index on semen parameters and reproductive hormones in human males: a systematic review with meta-analysis. Human Reproduction Update.2010; 16: 293–311.
8. World Health Organization, 2012.Global database on body mass index.
9. Cooper TG, Noonan E et al. World Health Organization reference values for human semen characteristics. Human Reproduction Update.2010; 16(3): 231-245.
10. Hammoud AO, Gibson M.Impact of male obesity on infertility: a critical review of the current literature. Fertility 2008; 90(4): 897-904.
11. Calogero A, Polosa Ret al. Cigarette smoke extract immobilizes human spermatozoa and induces sperm apoptosis.2009; 19(4):564-71.
12. Rakesh Sharma, Kelly R Biedenharn, Jennifer M Fedor.Lifestyle factors and reproductive health: taking control of your fertility.2013; 11: 66.
13. Sermondade C. Faure L. Fezeu.BMI in relation to sperm count: an updated systematic review and collaborative meta-analysis. Human Reproduction Update. 2013; 19(3):221 – 231.
14. Smit M, Romijn JC, Sperm chromatin structure is associated with the quality of spermatogenesis in infertile patients. 2010; 12(4): 480–489.
15.Nicole O. Palmer, Hassan W. Bakos et al. Impact of obesity on male fertility, sperm function and molecular composition. 2012; 2(4): 253–263.
16.Pauli EM, Legro RS, Demers LM et al. Diminished paternity and gonadal function with increasing obesity in men.2018; 16(4): 285–290.
17. Belloc S, Cohen Bacrie Metal. High body mass index has a deleterious effect on semen parameters except morphology: results from a large cohort study. Fertility and Sterility.2014; 102(5):1268–127314.
18. OboseRufus,Osaikhuwuomwan James. Male obesity and semen quality: Any association. 2018; 16(4): 285–290.
19. Bullen V, Judge S. The impact of obesity on male fertility. 2015; 1(3): 99 –107.
20. Teerds KJ, Keijer J. Functional relationship between obesity and male reproduction: from humans to animal models.2011; 17(5):667-83.
21. K. K. Shukla. Recent scenario of obesity and male fertility.2014; 2: 809–818.
