A study on the quality of life in children and adolescents with type I Diabetes Mellitus
Rajapriya S¹, Lathadevi GV², Meena Kumari M³
¹III Year Post Graduate, ²Professor, Department of Physiology, ³Consultant Paediatric endocrinologist, Department of Endocrinology,
PSG Institute of Medical Science & Research, Coimbatore, Tamil Nadu, India
Abstract
Background: Diabetes mellitus is expected to increase from 425 million in 2017 to 629 million by 2045 worldwide.Diabetes mellitus diagnosed during childhood presents an increased risk of complications in an early and productive stage of life, leading to a reduction in mean life expectancy of 10 to 20 years which is especially prevalent in developing countries. Children and adolescents with Type 1 diabetes Mellitus are at a heightened risk for mental health issues, including diabetes distress, depression, anxiety, and disordered eating. Health-Related Quality of Life (HRQOL) is an essential health outcome in clinical trials and healthcare.
Aim: To evaluate the quality of life in children and adolescents with Type 1 Diabetes Mellitus for an effective management using Pediatric Quality of Life Inventory (PedsQL) tool questionnaire.
Materials and Methodology: This was a prospective study. After ethical approval, 60 subjects who qualify the inclusion criteria were enrolled. The PedsQL tool questionnaire were given separately to child and parent. The child self-report and parent proxy-report were obtained and the PedsQL 4.0 Generic Core Scales were calculated. Statistical analysis was done using paired student ‘t’ test.
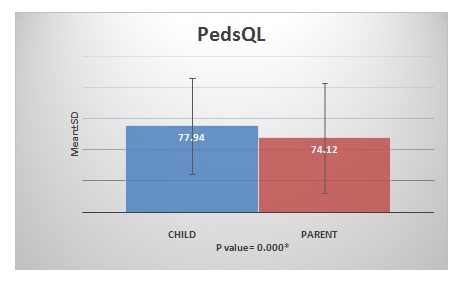
Results: The PedsQL score of children was 77.94 ± 15.59 and that of parents was 74.12 ± 17.78. There was a significant difference between the child’s self-report and the parents’- proxy report. The p value was statistically significant (p =0.000).
Conclusion: These findings emphasizes the importance of the psychosocial assessment and regular counselling to the children and adolescents with Type 1 DM which is a chronic disease along with their routine medical treatment which insists on good glycemic control.
Keywords: type1 diabetes mellitus, HRQOL, PedsQL
Corresponding Author ::
Dr.Rajapriya S, III Year Post Graduate,
MD, Physiology,
B-104, Sneham Apartments,
Anna Nagar, Peelamedu,
Coimbatore – 641004.
Telephone: +91 9445228598
E-mail: [email protected]
Introduction: Diabetes mellitus (DM) is one of the most common metabolic diseases worldwide. Its prevalence has increased in the past decade. Almost 90% of cases classified as Type 1 DM, with an autoimmune cause are children aged under 15 years. It represents one of the major chronic disease in pediatric age group. The Diabetes Atlas 2017 estimates that there are 128,500 children and adolescents with diabetes in India.
Type 1 DM, formerly known as juvenile or insulin-dependent diabetes is characterized by deficient insulin production.Symptoms include excessive thirst (polydipsia), constant hunger (polyphagia) excessive excretion of urine (polyuria), fatigue, weight loss and visual defects. It requires daily and lifelong insulin administration.
DM diagnosed during childhood presents an increased risk of complications in an early and productive stage. It leads to a reduction in mean life expectancy of 10 to 20 years, which is especially prevalent in developing countries like India.
Prospective clinical studies have clearly shown that a strict glycemic control from disease onset can delayor even prevent the onset of DM-related chronic vascular complications. However, strategies to prevent severe recurrent nocturnal hypoglycemia must also bedeveloped. This shows that the daily management of Type 1 DM has numerous challenges with it, to achieve a satisfactory metabolic control. It requires complex treatment through multiple insulin injections, periodic self-monitoring of blood glucose, strict mealtime schedule,regular physical exercise and frequent communication with health-care professionals.7,8 All these will impact the quality of life, especially of the children and adolescents.
Health-Related Quality of Life (HRQOL) is an essential health outcome in clinical trials and healthcare. The various aspects of health addressed by HRQOL are specific to each stage of cognitive development in children. It can reveal problems that even parents fail to notice. Thus, a standardized HRQOL assessment instrument is highly useful for detecting physical and emotional concerns from the perspectives of both children and caregivers.
The Pediatric Quality of Life Inventory (PedsQL) measurement model was designed to integrate the merits of generic and disease-specific instruments. The PedsQL 4.0 Generic Core Scales distinguish between healthy children and pediatric patients with acute or chronic health conditions and they have demonstrated sensitivity, responsiveness andan impact on clinical decision-making.
The Pediatric Quality of Life Inventory (PedsQL) is a modular instrument designed to measure health-related quality of life (HRQOL) in children and adolescents aged 2–18 years. The PedsQL 4.0 Generic Core Scales are child self-report and parent proxy-report scales developed as the generic core measure to be integrated with the PedsQL disease-specific modules.
Aim: To evaluate the quality of life in children and adolescents with Type 1 Diabetes Mellitus for an effective management.
Objectives: To assess the health-related quality of life (HRQOL) in children and adolescents with Type 1 Diabetes Mellitus aged 2–18 years, using Pediatric Quality of Life Inventory (PedsQL) tool which is based on the physical and emotional concerns of both children and their caregivers, to improve the diabetic care to the child and to restore their psychological wellness.
Study design: Prospective study.
Place of study: This study was done in the Department of Endocrinology.
Study population: 60 Type 1 Diabetes Mellitus children and adolescents and their caregivers.
Study period: March- May 2019.
Inclusion criteria:
1. Type 1 Diabetics.
2. Sex- both girls and boys.
3. Age group-between 2-18 years.
4. Parents or caregivers of Type 1 DM children.
Exclusion criteria:
1. Age group > 18 years.
2. Type 2 Diabetics.
Methodology: After obtaining ethical clearance from the Institutional Human Ethics Committee,60 subjects who qualify the inclusion criteria were enrolled. Informed consents were obtained from the parent as well as the children of age above 13 years. For children below 13 years of age a verbal consent was obtained in the presence of the parents. The children and adolescents with Type I Diabetes Mellitus who were on regular follow up to the Endocrinology OPD were taken for the study. Pediatric Quality of Life Inventory (PedsQL) tool questionnaires were given separately to the child and parent after proper instructions and guidance. Parents who were illiterate were guided by paramedical student volunteers.
The PedsQL 4.0 generic core scales:
The Child and Parent Reports of the PedsQL TM 4.0 Generic Core Scales for:
– Toddlers
– Young Children (ages 5-7),
– Children (ages 8-12),
– Teens (ages 13-18),
are composed of 23 items comprising 4 dimensions.
Description of the questionnaire:
The 23-item PedsQL 4.0 Generic Core Scales encompass:
1) physical functioning (8 items),
2) emotional functioning(5items),
3) social functioning (5 items),and
4) school functioning (5 items).
Child self-report includes ages 5–18years, and parents’ proxy-report includesages 2–18 years. Though there were different sets of questionnaires for different age groups, the items for each form were essentially identical. It was based on the problems they had during the past 1 month.
Scoring of dimensions:
1) Item Scaling:
-For Children and adolescents (ages 8-18) report and for all parent’s report.
5-point scale: From 0-4
0 (Never), 1 (Almost Never), 2 (Sometimes), 3 (Often) and 4 (Almost always)
-For the Young Child (ages 5-7) child report
3-point scale: 0,2 and 4.
0 (Not at all), 2 (Sometimes) and 4 (A lot)
2) There was no separate weighing of items done.
3) Extension of the Scoring Scale-Scores were transformed on a scale from 0 to 100.
4) Scoring Procedure
Step 1: Transform Score
Items were reverse scored and linearly transformed to a 0-100 scale as follows:
0=100, 1=75, 2=50, 3=25, 4=0.
Step 2: Calculate Scores
Score by Dimensions:
-If more than 50% of the items in the scale were missing, the scale scoreswere not computed,
-Mean score = Sum of the items over the number of items answered.
Statistical analysis
SPSS (Statistical Package for the Social Science) software version- 24, Chicago, Illinois was used for statistical analysis of values obtained. The data collected were analyzed usingpaired student-t test
Physical Health Summary Score = Physical Functioning Scale Score
Psycho social Health Summary Score = Emotional + Social + School Functioning Scores.
Total Score= Sum of all the items over the number of items answered on all the Scales.
Results
The children and their parents or caregivers gave 100% response rate. In some instances, the children reported lower scores than the parents and many instances the parents’ proxy score was lower than their child’s score. The physical score was slightly higher than the psychosocial score in the children’self-report. Whereas, the psychosocial score was higher than the physical score in the parent’s proxy report. The mean value of PedsQL score of children was 77.94 ± 15.59 and that of parents was 74.12 ± 17.78.
There was a significant difference between the child’s self-report and the parents- proxy report. The p value was statistically significant (p =0.000). Overall, parents consistently reported lower scores for their children HRQoL than children reported themselves.
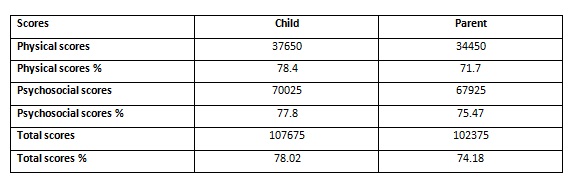
Comparison of PedsQL 4.0 Generic Core Scale scores between children self report and parent proxy report.
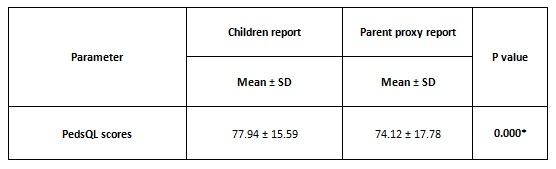
*P-value < 0.005 – statistically significant
Discussion: Type 1 Diabetes contributes to the development of problems in physical, social, academic activities and psychological functioning involving the parent-child relationship and can negatively affect their quality of life. The role of family and friends to their physical wellbeing and emotional support is also crucial.
In our study there was a significant difference in the PedsQLscores of parent and children.
Physical scores:
Overall, the children reported better physical scores in contradiction to the parents’report, which showed lower physical scores than psychosocial scores. This could be due to parental perception of the challenges that are associated with lifestyle modifications for their child. It also reflects their concern for the short and long-term complications of diabetes which their children may have.
Psycho social scores: Overall, the parents’ reported lower psycho social scores than their children.
In some instances, the children’s scores were lower than the parents, which suggest that the parents have not noticed the difficulties that their children had in their psycho social activities or may be associated with a negative impact on parents’ psychological well-being or that they overestimated their child’s ability.
In many instances, the parents’ proxy psycho social score was lower than their child’s score. It canbe due to a lack of communication and understanding between the parent and children or may be due to the stigma, which the parents may have in the ability of their children with Type 1 DM.
Total scores: Overall, parents consistently reported lower PedsQL scores for their children HRQoL than children reported themselves.
The current medical practice tends to primarily focus on achieving metabolic control, concentrating on the biomarkers of the disease. Less emphasis is laid on addressing the psychosocial components of disease management and the impact it has on children and adolescents’ HRQoL. Diabetes management must be tailored to meet specific challenges faced by children, adolescents and their families.
Sometimes, adolescence is considered as a stressful time in the life of teens because of the major developmental, hormonal and psychological changes that occur. T1DM, which involves multiple lifestyle and psychosocial changes can further comprehend to an already dynamic period.17Systematic review evidence suggests that specialized adolescent health clinics or “transition clinics” are successful in improving many health outcomes associated with diabetes in adolescence.
Regular counselling can be conducted to bring down the differences in between the PedsQL scores of the children and their parents.
The Department of Peadiatric endocrinology in our institution has taken a call over this situation to bring the children and their parents out of their stigma on Type 1 DM. By regularly conducting camps, awareness program and by exposing children to people who have succeeded inspite of having Type 1 DM such as academic toppers, marathon runners, hill climbers, swimmers,etc., can help them accept their medical condition and go through the challenges that come with it, boldly. It helps them to face their life with confidence. This way it improves their quality of life and thereby promotes them to become a responsible youth in the society in future.
Study limitations and recommendations: The sample size taken for the study was small and it can be increased in the future studies. The relationship of age, gender, glycemic status, socio economic status and literacy rate among the parents with the PedsQL scores were not considered in our study. This psychosocial assessment can be done periodically during each visit to monitor their psychosocial well-being as well as to assess the effectiveness of the camps and awareness program. Future studies could assess the effectiveness of involving children and adolescent mental health experts in improving the HRQoL of children, adolescents as well as their parents.
Conclusion: These findings emphasizes the importance of the psychosocial assessment and regular counselling to the children and adolescents with Type 1 DM which is a chronic disease along with their routine medical treatment which insists on the glycemic control.The importance of a healthy parent- children communication has to be born in mind while working towards improvement in psychological wellbeing along with an optimal metabolic control.
Acknowledgements: Nil.
Conflict of Interest: Nil.
References:
1. International Diabetes Federation. IDF Diabetes Atlas, 8thedn. Brussels, Belgium: International Diabetes Federation, 2017. Available from: http://www.diabetesatlas.org.Accessed February 17, 2019.
2. Patterson C, Guariguata L, Dahlquist G, Soltész G, Ogle G, SilinkM. Diabetes in the young -a global view and worldwide estimates of numbers of children with type 1 diabetes. International Journal of Diabetes Research and Clinical Practice. 2014;103:161-75.
3. Diabetes Control and Complications Trial Research Group,Nathan DM, Genuth S, Lachin J, Cleary P, CroffordO,et al. The effect of intensive treatment of diabetes onthe development and progression of long-term complicationsin insulin-dependent diabetes mellitus. New England Journal of Medicine.1993;329:977-86.
4. White NH, Cleary PA, Dahms W, Goldstein D, MaloneJ, Tamborlane WV. Diabetes Control and ComplicationsTrial (DCCT)/Epidemiology of Diabetes Interventions andComplications (EDIC) Research Group. Beneficial effects ofintensive therapy of diabetes during adolescence: outcomesafter the conclusion of the diabetes control and complicationstrial (DCCT). Journal of Pediatrics. 2001;139:804-12.
5. Rewers MJ, Pillay K, de Beaufort C, Craig ME, Hanas R, AceriniCL, et al. Assessment and monitoring of glycemic controlin children and adolescents with diabetes. Journal of Pediatrics Diabetes.2014;15:102-14.
6. American Diabetes Association. Children and adolescents Diabetes Care. 2016;39:S86-93
7. Guttmann-Bauman I, Flaherty BP, Strugger M, McEvoy RC.Metabolic control and quality of life self-assessment in adolescents with IDDM. Diabetes Care. 1998;21:915-8.
8. Jaser SS, Faulkner MS, Whittemore R, Jeon S, Murphy K,Delamater A, et al. Coping, self-management and adaptation in adolescents with type 1 diabetes. Annals of Behavioral Medicine.2012;43:311-9.
9. Rubin RR. Quality of life and diabetes. Diabetes Metabolism Research and Reviews.1999;15:205-18.
10. Reliability and validity of a diabetes quality-of-life measure for the diabetes control and complications trial (DCCT). The DCCTResearch Group. Diabetes Care. 1988;11:725-32.
11. Varni JW, Burwinkle TM, Lane MM. Health-related quality of life measurement in pediatric clinical practice: an appraisal andprecept for future research and application. Health Quality Life Outcomes. 2005;3:34.11.
12. Johnson SB, Perwien AR: Insulin-dependent diabetes mellitus. In Quality of Life in Child and Adolescent Illness: Concepts,Methods, and Findings. Koot HM, Wallander JL, Eds. East Sussex, UK, Brunner-Routledge, 2001, p. 373–401.
13. Varni JW, Seid M, Rode CA: The PedsQL: measurement model for the Pediatric Quality of Life Inventory. Medical Care, 1999;37:126–139.
14. Varni JW, Seid M, Kurtin PS: The PedsQL 4.0: reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Medical Care, 2001;39:800 – 812.
15. Varni JW, Seid M, Knight TS, Burwinkle TM, Brown J, Szer IS: The PedsQL in pediatric rheumatology: reliability, validity, and responsiveness of the Pediatric Quality of Life Inventory Generic Core Scales and Rheumatology Module. Journal of Arthritis Rheumatology , 2002;46:714 –725.
16. F. AlBuhairan, A. Harrison, M. Kaufman, R. AreemitAdolescent psychosocial development and evaluation: global perspectives INTECH Open Access Publisher (2012).
17. T. Wysocki, A. Lochrie, H. Antal, L.M. Buckloh,Youth and parent knowledge and communication about major complications of type 1 diabetes: associations with diabetes outcomes,Diabetes Care, 34 .August 8,2011, pp. 1701-1705.
18. R. Crowley, I. Wolfe, K. Lock, M. McKee. Improving the transition between paediatric and adult healthcare: a systematic reviewArch Dis Child, 96 (Jun (6)) (2011), pp. 548-553.
