Assessment of autonomic status and neutrophil lymphocyte ratio in type 2 diabetics and its
correlation with their disease duration
Vishnu Priya K¹, Anitha A², Narmadha Lakshmi R³, Ravi S4, Getrude Banumathi P5
¹II Year Post-graduate, ²Professor, Department of Physiology, ³Department of Internal Medicine, 4Department of Pathology, 5Associate Professor, Department of Community Medicine, Chengalpattu Medical College, Chengalpattu, Tamil Nadu, India
Abstract
Background: Cardiac Autonomic Neuropathy (CAN) is a significant cause of morbidity and mortality in Type 2 Diabetes Mellitus (DM). Association between Diabetes Mellitus and cardiovascular status using Heart rate variability (HRV) has been shown in many studies. Chronic inflammation plays a central role in the development, progression and pathogenesis of Type 2 Diabetes Mellitus and particularly leucocytes play a key role in it. Neutrophil Lymphocyte Ratio (NLR) is a marker in acute coronary syndrome, Stroke and in Ulcerative colitis. With this background, HRV and NLR were assessed in Type 2 DM.
Aim: To correlate the Autonomic status and Neutrophil Lymphocyte ratio with Duration of Type 2 DM.
Materials and methods: A cross-sectional study was done after Institutional Ethics Committee approval, with written informed consent on 135 Type 2 DM patients of age 30 to 60 years of both Gender.3 groups were taken based on their disease duration (in years) as A – 1 to 5, B – 6 to 10, C – More than 10. Complete Blood Count was done by Automated SysmacAnalyser and NLR was calculated. Autonomic status was assessed by the Compass 31 Questionnaire and HRV and data analyzed using SPSS 21.0.
Results: Progressive increase in mean NLR, mean LF nu, mean LF/HF ratio was seen from Group A to C. “p-value” less than 0.05 was taken as significant. Correlation between NLR and LF/HF ratio(r =0.249) showed a Positive association.
Conclusion: NLR can also be used as a screening tool in Type 2 DM at an earlier stage for further assessing Cardiac Autonomic Neuropathy (CAN).
Keywords: cardiac autonomic neuropathy, heart rate variability, neutrophil lymphocyte ratio, type2DM.
Corresponding Author ::
Dr. Anitha Achuthan, Professor,
Department of Physiology,
Chengalpattu Medical College,
Chengalpatttu, Tamil Nadu, India
Telephone: +91 9443154246
E-mail: [email protected]
Introduction: Cardiac Autonomic Neuropathy is an often underdiagnosed microvascular complication in Type 2 DM. Annual incidence of Cardiac Autonomic Neuropathy is about 2% in Diabetes mellitus. It can occur from 32% to 73% in Diabetic patients. Many studies have showed association between Diabetes and cardiovascular problems using Short term Heart rate Variability. ¹Vinik et.al, says that there is activation of inflammatory cytokines in Diabetic patients and they correlate with abnormalities in sympathovagal balance. They further added that dysfunction of the autonomic system predicts cardiovascular risk and sudden death in patients with Type 2 Diabetes. Polyol pathway, oxidative stress has leading roles in cardiac autonomic neuropathy. ³Heart rate variability is an earliest indicator and most frequent finding in asymptomatic cardiovascular autonomic dysfunction³.
There are several studies which portray the link between systemic inflammation and cardiac problems in Diabetes. Neutrophils are active non- specific inflammatory mediator initiating the first line of defense and Lymphocytes are regulatory or protective component of inflammation.3 Neutrophil Lymphocyte Ratio is a measure of systemic inflammation in relation to Diabetes. Systemic inflammation measured by NLR has significant association with prevalent chronic conditions such as Diabetes and Hypertension. Studies have reported Neutrophil Lymphocyte Ratio as a marker in Metabolic syndrome, as well as in Prediabetes. ATTICA study showed that there was a positive association between low-grade inflammation and diabetes without any evidence of cardiovascular disease. Hematological parameters are assessed as markers for inflammation in Diabetes nowadays. Hence the aim of present study was to assess autonomic status by Compass 31 questionnaire and short term Heart rate variability, estimate Neutrophil Lymphocyte ratio and correlate these with duration of disease.
Methodology: This was a cross sectional study done in Chengalpattu Medical College.Informed written consent was obtained from all patients after getting approval from Institutional ethics Committee.Rules of modified Helsinki’s declaration were followed. 135 Type 2 Diabetic patients of gender matched were chosen from Outpatient department of Internal Medicine. They were divided into 45 subjects with 1-5 years of duration of disease as group A, 45 subjects with 6-10 years of duration as group B, 45 subjects with more than 10 years of duration as group C. Patients of age 30 to 60, Type 2 DM, patients co existing with Hypertension, Patients on Oral Hypoglycaemic agents or Insulin or both were included in the study. Patients of Type 1 DM, Patients with infections, acute complications, Coronary artery disease, Patients on oral steroids, Smokers and Alcoholics were excluded from the study.
Blood Count was done by Automated SysmacAnalyser and Neutrophil Lymphocyte Ratio was calculated from percentage of neutrophils and lymphocytes. Autonomic status was assessed by Compass 31 Questionnaire by translating the questionnaire in native language and elicited scores in front of a third person. Short term HRV analysis was performed by recording Lead II ECG for five minutes in quiet room using Variowin software(HR 2.0) at room temperature of 24-26 degree in NCD Clinic, Chengalpattu Medical College. Patients, after emptying the bladder were made to feel relax before starting the recording of HRV. Patients were asked to avoid beverages which stimulates sympathetic system and drugs which have effect on ANS were withdrawn overnight. HRV parameters were analysed using SPSS 21.0 version.
Results: Results of three groups were analyzed by ANOVA, Chi – Square test, Spearman Correlation test using (IBM) SPSS software -21.0. There were no significant statistical differences in terms of sex, in three groups. In all the three groups, 21 were male and 24 female patients. 10 persons in Group A, 13 persons in Group Band 28 persons in Group Cwere having both Diabetes and Hypertension. p<0.05 was taken as significant. Progressive increase of mean NLR was seen from group A to group C. (1.48±0.68,1.78±0.64,2.37±0.71,p value-0.00001) as shown in table no: 2. NLR more than 2 was taken as abnormal.
Table no: 1 shows the Baseline Characteristics
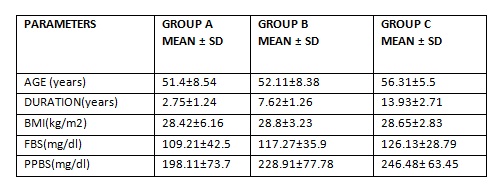
Table no: 2 Parameters that are analysed with ANOVA
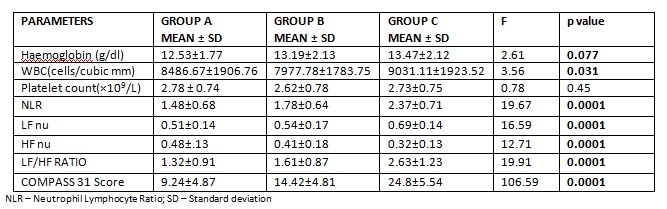
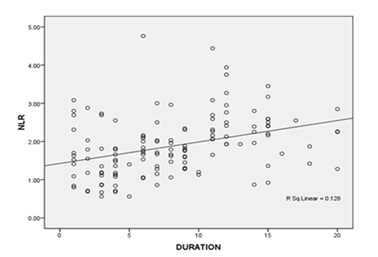
Fig no: 1 shows Positive association between NLR and Duration of DM.(r value =.411**)
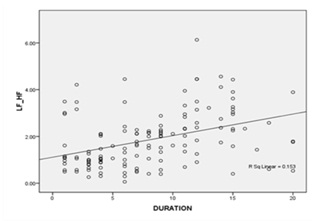
Fig no: 2 shows Positive association between LF/HF RATIO and Duration of DM. (r value = .430**)
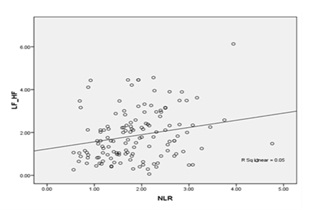
Fig no: 3 shows Positive association between LF/HF RATIO and NLR.(r value = .249**)
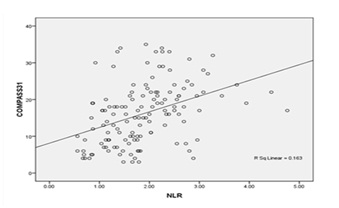
Fig no: 4 shows Positive association between COMPASS 31 SCORE and NLR.(r value =.467**)
Discussion: In current scenario, incidence of Myocardial Infarction and Coronary Artery Disease (21.2%) in Diabetic patients is high and is the leading cause of death. Verma et al describes that Cardiac autonomic regulation assessed by heart rate recovery and heart rate variability was impaired in diabetic patients.
Cardiac autonomic neuropathy and DM: Benichou et al in their meta-analysis quoted that T2DM patients exhibited a strong decrease in HRV. Both sympathetic and parasympathetic activity were decreased compared with non–T2DM patients, which can be explained by the deleterious metabolic effects of blood glucose levels on autonomic system. Diabetes has a negative influence on almost all HRV parameters, reflecting the fact that diabetes leads to a cardiac autonomic dysfunction. Our study shows results similar to these previous studies as Mean LF nu, Mean LF/HF Ratio increases as the duration increases and Mean HF nu decreases as the duration increases.
Inflammation and DM: Gou et al conducted a study in Chinese population and proposed that increased NLR was associated with prevalence and incidence of DM and added that baseline NLR can be a better predictor and a marker in diabetes.
Verdoia et al says that Neutrophil Lymphocyte ratio has emerged as a potential marker for vascular complications in Diabetes and they correlated it with the outcome of coronary artery disease in DM.
Autonomic symptoms were also progressively more in regard to duration of diabetes. Compass 31 score is a questionnaire involving 6 domains namely Orthostatic Intolerance, Vasomotor, Secretomotor, Gastrointestinal, Bladder, Pupillomotor which covers autonomic symptoms to be elicited in patients. The score ranges from 0 to 100.12Our study tried to link the complaints of the patients with the evolving changes in vasculature and heart. Though there is a profound subjective component in the questionnaire, yet it supported our study results.
Link between neutrophil lymphocyte ratio and cardiac autonomic neuropathy: Pathophysiology of diabetes is due to Hyperglycemia and this causes altered glucose metabolism. This in turn leads to the production of AGEP – Advanced Glycation End Products, which once formed is irreversible. This causes ischemic death of neurons and causes initially vagal denervation, followed by increase in sympathetic tone and finally patients land up with sympathetic overactivity. Vagus nerve is earlier involved in autonomic neuropathy because of its long course. Parasympathetic fibers being the longest fibers are affected first due to atherosclerotic changes of vasa nervorum. Further cluster of metabolic and hemodynamic abnormalities, including a disadvantageous lipid profile and altered diurnal blood pressure rhythm accelerates this denervation.
On the other hand, endothelial dysfunction causes neutrophilia because of inflammation and atherosclerosis. There is recruitment of neutrophils at the inflammatory site as it is the first line of defense, it promotes further scavenging effect by macrophages. One might wonder that in chronic inflammation like Diabetes, one would expect a lymphocytosis. But, in these patients there is decreased expression of IL 2 receptors and increased apoptosis thereby causing Lymphopenia. This can be attributed for increase in NLR in these patients.This hypothesis is in concordance with our study results as we obtained an increased neutrophil count and decreased lymphocyte count and an increased NLR in our study group.
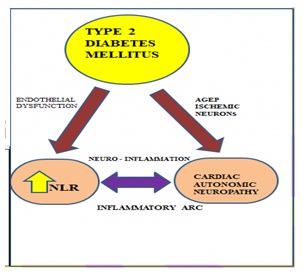
Fig no: 5 shows the relationship between Neutrophil Lymphocyte Ratio, Cardiac Autonomic Neuropathy in Type 2 Diabetes Mellitus. AGEP – Advanced Glycation End Products
Watkins et al. says that sensory neurons can sense infection/injury and the ongoing inflammation. The responses to interleukin (IL)-1 induced inflammation are mediated by the vagus nerve, and could be abolished by vagotomy or a selective competitive antagonist of the IL-1 receptor. Oxygen, Glucose and other metabolites are sensed by specialized glomuscells in association with the autonomic nervous system, and on activation release Dopamine and Norepinephrine. They further narrated a hypothesis involving Vagus nerve and splenic nerve which are components of autonomic nervous system in an inflammatory arc responding for exogenous (e.g. pathogen-associated molecular pattern [PAMPs]) or endogenous (e.g. damage-associated molecular pattern [DAMPs]) molecules. This leads to afferent Action potentials being transmitted along the vagus nerve to the brainstem and other nuclei that control outgoing signals in the vagus nerve. Increased activity in the vagus nerve to the spleen and other organs reduces the innate immune system’s response to DAMPs and PAMPs, decreases the release of cytokines, and suppresses inflammation by triggering the binding of anti – inflammatory cytokines to receptors in macrophages and this heralds the inflammation. But in diabetes, what happens is early vagal denervation, which could not break the inflammatory arc thus promoting inflammation.
Pavlov and Tracey had done a work contributing a link between immunity and vagus nerve which supports our study. They say that cholinergic suppression can alleviate the symptoms associated with metabolic syndrome and type 2 Diabetes mellitus.15But the molecular mechanisms for downregulating at receptor levels is a grey area till now. Further, NLR is a proven neuro -inflammatory marker. So, inflammation and Cardiac Autonomic Neuropathy occurs simultaneously in Diabetic patients and worsens the condition of the patients.
Thayer describes, an association between heart rate variability and inflammation may support the concept of a cholinergic anti-inflammatory pathway. According to him, Neural mechanisms also regulate inflammation; Vagus nerve activity inhibits macrophage activation and the synthesis of tumor necrosis factor in the reticuloendothelial system through the release of acetylcholine. Our study also sticks on this Neuro – inflammatory pathway in Cardiac autonomic neuropathy in DM because we found that patients with abnormally low HRV had an increased NLR in our study group.
Although cholinergic suppression of inflammation can contribute specifically to alleviate metabolic complications in metabolic syndrome and type 2 diabetes mellitus, these complex interactions and the contribution of central and peripheral mechanisms in this regulation are topics of ongoing study across the world till now.Additionally, intracellular mechanisms by which cholinergic signals control obesity-associated inflammation and modulate insulin signaling are under investigation.
Lorenzo et al showed that there was a significant relationship between Diabetes and White blood cell count and abnormal Neutrophil Lymphocyte ratio obtained in their study should have a pathogenesis other than obesity and insulin resistance.17This correlates with our study results because our study participants were comparable with their BMI. Shiny et al in their study in Indian population have said that NLR increases as severity of glucose intolerance increases.They have pointed out that Hyperglycemia reduces the apoptosis of neutrophils and thus promotes endothelial dysfunction.
Kudat et al pointed out the relation between decreased heart rate variability and the presence of microvascular complications and they added that Insulin therapy should be started at the earliest to prevent cardiac autonomic dysfunction.
Hussain et al proposed that increased NLR has association with poor glycemic control, elevated HbA1C and CRP and further added that NLR can be used as a disease monitoring tool in follow up of DM patients as it is a direct cause of cardiovascular disease in DM like CRP.
Lou et al described that NLR has a significant correlation with insulin resistance in Newly diagnosed Diabetic patients.3 This stresses our point of importance of screening diabetic patients at the earliest.
Conclusion: Inflammation and Cardiac Autonomic Neuropathy occurs simultaneously in Diabetes, as the duration increases and routine screening of them is necessary as Diabetes is the third leading cause of death.
Limitations of the study: HbA1C and CRP was not done because of cost effectiveness. Correlation of drug effect was also not done. Lipid profile if taken would have added a point towards cardiovascular risk in patients.
Implications: NLR is cost effective and can be easily performed. It should be recommended in Diabetic patients in their routine complication screening protocol once in 6 months and patients with abnormal NLR value should be subjected to Short term Heart Rate Variability analysis for screening of sympathovagal balance. This can throw an idea on persons who are at the start of complications so that effective measures can be taken in order to prevent these complications and thus improving quality of life in these patients.
Future scope of the study: NLR and HRV estimation in newly diagnosed diabetic people can clinch the incidence of cardiac problems at the earliest.
Acknowledgments: The authors are thankful to Dr.S. Sujatha, MD(SPM), Assistant Professor, Department of Community Medicine, Chengalpattu Medical College and Hospital, Chengalpattu for her help in statistics. At this moment, we thank all our study people.
Conflict of Interest: Nil.
References:
1. Babu HRH, Muralikrishnan K. Frequency Domain Analysis of Heart Rate Variability in Type II Diabetes Mellitus Patients in a Tertiary Care Hospital.:5.
2. Vinik AI, Erbas T, Casellini CM. Diabetic cardiac autonomic neuropathy, inflammation and cardiovascular disease. J Diabetes Investig. 2013 Jan;4(1):4–18.
3. Lou M, Luo P, Tang R, Peng Y, Yu S, Huang W, et al. Relationship between neutrophil-lymphocyte ratio and insulin resistance in newly diagnosed type 2 diabetes mellitus patients. BMC Endocr Disord. 2015 Dec;15(1):9.
4. Imtiaz F, Shafique K, Mirza S, Ayoob Z, Vart P, Rao S. Neutrophil lymphocyte ratio as a measure of systemic inflammation in prevalent chronic diseases in Asian population. Int Arch Med. 2012;5(1):2.
5. Buyukkaya E, Karakaş MF, Karakaş E, Akçay AB, Tanboga IH, Kurt M, et al. Correlation of Neutrophil to Lymphocyte Ratio With the Presence and Severity of Metabolic Syndrome. Clin Appl Thromb. 2014 Mar;20(2):159–63.
6. Mertoglu C, Gunay M. Neutrophil-Lymphocyte ratio and Platelet-Lymphocyte ratio as useful predictive markers of prediabetes and diabetes mellitus. Diabetes Metab Syndr Clin Res Rev. 2017 Nov;11:S127–31.
7. Pitsavos C, Tampourlou M, Panagiotakos DB, Skoumas Y, Chrysohoou C, Nomikos T, et al. Association Between Low-Grade Systemic Inflammation and Type 2 Diabetes Mellitus Among Men and Women from the ATTICA Study. Rev Diabet Stud. 2007;4(2):98–104.
8. Verma S, Alam R, Ahmad I, Singla D, Ali K, Hussain ME. Effect of glycemic control and disease duration on cardiac autonomic function and oxidative stress in type 2 diabetes mellitus. J Diabetes Metab Disord. 2018 Dec;17(2):149–58.
9. Benichou T, Pereira B, Mermillod M, Tauveron I, Pfabigan D, Maqdasy S. Heart rate variability in type 2 diabetes mellitus: A systematic review and meta– analysis. :19.
10. Guo X, Zhang S, Zhang Q, Liu L, Wu H, Du H, et al. Neutrophil:lymphocyte ratio is positively related to type 2 diabetes in a large-scale adult population: a Tianjin Chronic Low-Grade Systemic Inflammation and Health cohort study. Eur J Endocrinol. 2015 Aug;173(2):217–25.
11. Verdoia M, Schaffer A, Barbieri L, Aimaretti G, Marino P, Sinigaglia F, et al. Impact of diabetes on neutrophil-to-lymphocyte ratio and its relationship to coronary artery disease. Diabetes Metab. 2015 Sep;41(4):304–11.
12. Sletten DM, Suarez GA, Low PA, Mandrekar J, Singer W. COMPASS 31: A Refined and Abbreviated Composite Autonomic Symptom Score. Mayo Clin Proc. 2012 Dec;87(12):1196–201.
13. Kumari KP, Neeraja M, Kumar SC, et al. Cardiovascular Parasympathetic Changes in Type-II Diabetic Mellitus. Int J Integr Med Sci. 2016 Aug 31;3(8):374–83.
14. Nurlaelatiningsih C, Sastradimaja SS, Defi IR. Relationship between Neutrophil-Lymphocyte Ratio and Cardiac Autonomic Neuropathy in Diabetes Mellitus Type 2 Patients. International Journal of Integrated Health Sciences. 2017 Mar 31;5(1):15-20.
15. Pavlov VA, Tracey KJ. The vagus nerve and the inflammatory reflex—linking immunity and metabolism. Nat Rev Endocrinol. 2012 Dec;8(12):743–54.
16. Thayer JF. Vagal tone and the inflammatory reflex. Cleve Clin J Med. 2009 Apr 1;76(Suppl_2):S23–6.
17. Lorenzo C, Hanley AJ, Haffner SM. Differential white cell count and incident type 2 diabetes: the Insulin Resistance Atherosclerosis Study. Diabetologia. 2014 Jan;57(1):83–92.
18. Shiny A, Bibin YS, Shanthirani CS, Regin BS, Anjana RM, Balasubramanyam M, et al. Association of Neutrophil-Lymphocyte Ratio with Glucose Intolerance: An Indicator of Systemic Inflammation in Patients with Type 2 Diabetes. Diabetes Technol Ther. 2014 Aug;16(8):524–30.
19. Kudat H, Akkaya V, Sozen A, Salman S, Demirel S, Ozcan M, et al. Heart Rate Variability in Diabetes Patients. J Int Med Res. 2006 May;34(3):291–6.
20. Hussain M, Babar MZM, Akhtar L, Hussain MS. Neutrophil lymphocyte ratio (NLR): A well assessment tool of glycemic control in Type-2 diabetic patients.
